Titration
Titration is used to find the molarity of an acid or a base. In titration, an acid or base of known molarity is added to an acid or base of unknown molarity until neutralization occurs. Neutralization has occurred when the indicator has changed color. At that point, the molarity of H+ is equal to the molarity of OH-.
Molarity equation: MaVa=MbVb.
Ma= molarity of H+, Va= volume of acid, Mb=molarity of OH-, Vb= volume of base
Titration problems:
1. How many milliliters of 4.00 M NaOH are required to neutralize 50.0 milliliters of a 2.00 M solution of HNO3?
2. If 50.0 milliliters of KOH is needed to neutralize 200.0 milliliters of a .10 M solution of HNO3, what is the concentration of the acid?
Molarity equation: MaVa=MbVb.
Ma= molarity of H+, Va= volume of acid, Mb=molarity of OH-, Vb= volume of base
Titration problems:
1. How many milliliters of 4.00 M NaOH are required to neutralize 50.0 milliliters of a 2.00 M solution of HNO3?
2. If 50.0 milliliters of KOH is needed to neutralize 200.0 milliliters of a .10 M solution of HNO3, what is the concentration of the acid?
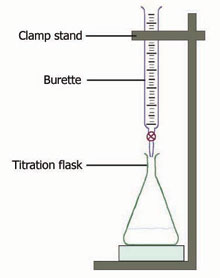
The ring stand holds the Burette in place. Place the flask directly under the Burette. The Burette is filled with acid and the flask is filled with base and indicator (Phenolphthalein). The tap on the Burette is turned and acid goes into the flask until the solution turns clear. This indicates that the solution is neutralized. Once the solution is neutralized, subtract the starting amount of acid from the ending amount of acid. Use the molarity equation, MaVa=MbVb to figure out the molarity of the unknown substance.
Procedure
1. Obtain and wear goggles. Tie back hair if applicable.
2. Rinse out two 100mL beakers, one 200mL flask, a Burette (with water and base) and a glass pipette (with water and acid).
3. Pour 10mL of each of the acid and the base, each in its own beaker.
4. Pour the acid into the Burette and pick up the base with the glass pipette. Put the base and three drop of Phenolphthalein into the flask.
5. Set up the apparatus at the left using the tips above.
Before taking the final exam, please take a moment out of your busy life and listen to this amusing and helpful song: /for-your-listening-pleasure.html
Procedure
1. Obtain and wear goggles. Tie back hair if applicable.
2. Rinse out two 100mL beakers, one 200mL flask, a Burette (with water and base) and a glass pipette (with water and acid).
3. Pour 10mL of each of the acid and the base, each in its own beaker.
4. Pour the acid into the Burette and pick up the base with the glass pipette. Put the base and three drop of Phenolphthalein into the flask.
5. Set up the apparatus at the left using the tips above.
Before taking the final exam, please take a moment out of your busy life and listen to this amusing and helpful song: /for-your-listening-pleasure.html

