pH and Indicators
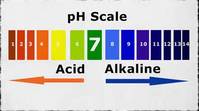
pH is calculated on a scale of 1-14. It indicates the strength of an acid or base.
pH<7 is an acid. The lower the number, the stronger the acid. 1-3 is considered a strong acid and 4-6 is considered a weak acid.
pH 7 indicates a neutral substance
pH>7 is a base. The higher the number, the stronger the base. 11-14 is considered a strong base and 8-10 is considered a weak base.
Stronger acids and bases will lose or gain a proton easier than a weaker one. They will also disassociate more in a water solution than a weak substance will.
Concentration is the amount of the dissolved substance in a solution.
pH<7 is an acid. The lower the number, the stronger the acid. 1-3 is considered a strong acid and 4-6 is considered a weak acid.
pH 7 indicates a neutral substance
pH>7 is a base. The higher the number, the stronger the base. 11-14 is considered a strong base and 8-10 is considered a weak base.
Stronger acids and bases will lose or gain a proton easier than a weaker one. They will also disassociate more in a water solution than a weak substance will.
Concentration is the amount of the dissolved substance in a solution.
Litmus Paper
Acid turns blue litmus paper red and red litmus paper red. Base turns blue litmus paper blue and red litmus paper blue. Neutral substances will not affect the color of litmus paper.
Phenolphthalein

An acid will turn phenolphthalein from pink to colorless. A base will turn it to a pinkish violet color. A neutral substance will make it pinkish clear.
Universal Indicator
Universal Indicator is an indicator that uses the pH scale. However, it uses colors in addition to numbers. The pH numbers are associated with corresponding colors: 1 - 3 is red, 3 - 6 is orange/yellow, 7 is green, 8 - 11 is blue and 11 - 14 is violet/purple.
Find out how to neutralize acids and bases: /neutralization.html