Buffers
- Buffers are aqueous (where water is the solvent) solutions that have a highly stable pH level. Buffers help to keep the pH from fluctuating, keeping the pH relatively constant even when an acid or base is added.
- Buffers consist of a weak acid and its conjugate base, or a weak base and its conjugate acid.
- Buffers are used for a variety of reasons:
- A buffer of carbonic acid (H2CO3) and its conjugate, hydrogen carbonate (HCO3-), work together to keep the pH of human blood at 7.4. Without the buffer keeping the pH close to 7.4, the pH would fluctuate and cause damage to the cells.
- Shampoo contains buffers of citric acid and its conjugate, sodium hydroxide, to balance out the natural alkalinity of soap, which would otherwise burn the scalp.
- Liquor manufacturers often add buffers to their mixes, which regulate pH levels to avoid spoilage of the alcohol.
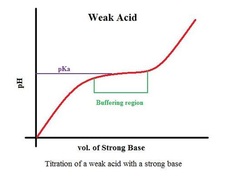
This is what a buffer consisting of a weak acid and strong base looks like. If you look at the buffer region, you’ll notice that adding more base will not change the pH significantly.
Now find out how to decipher if something is an acid or base by observing indicators: /ph-and-indicators.html
