Acids and Bases
Theories/definitions
- Svante Arrhenius - An acid is a substance that releases H+ ions in an aqueous solution (a solution in which water is the solvent) and a base is a substance that releases OH- ions in an aqueous solution.
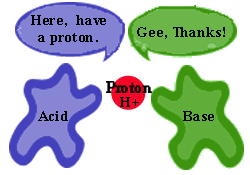
- Johannes Bronsted and T.M. Lowry (Bronsted-Lowry) - An acid is any particle that can donate a proton (H+) to another substance and a base is any particle that can accept a proton from another substance.
- G.N. Lewis - An acid is any particle that can accept a pair of electrons from another particle and a base is any particle that can donate a pair of electrons to another particle.
Properties
Acid
- tastes sour
- pH level is less than 7
- an electrolyte (conducts electricity when dissolved in water)
- neutralized by carbon dioxide
- turns blue litmus paper red
- neutralizes bases to form salt and water
Base
- tastes bitter
- pH greater than 7
- neutralizes acids to form salt and water
- turns red litmus paper blue
- feels slippery
- an electrolyte
If you want to know why you should care about acids and bases, here are real life examples: /uses-of-acids-and-bases.html